 This is called Dalton's atomic theory of gases. KE totally decides the temperature of the gas. Under this approximation, KE is the only term. As a convenience and a first approximation, gases are treated by ideal gas law. For our usual world, matter (gas, liguid or solid) doesn't reach that part of diagram. So, what happens to the left side of the potential energy diagram, where PE is overwhelmingly positive? That part of diagram is only used for understanding (or maybe for some extreme cases). As a result, the gas can no longer exist because it condenses to liquid or solid. When gas molecules get close enough as shown in the middle region of the diagram, the potential energy gets negative enough to cancel 100% of kinetic energy (which is always positive) and more, the total energy becomes negative. The (average) kinetic energy dominates and total energy is definitely positive. This is when gas molecules are far apart (large $r / \sigma$) and the gas state rules. On the right side shows the (average) potential energy of a gas which is always slightly negative. The diagram can roughly be divided into 3 regions. The average distance between the particles would be the distance between the centerpoints of two neighboring balls each with volume V/N. 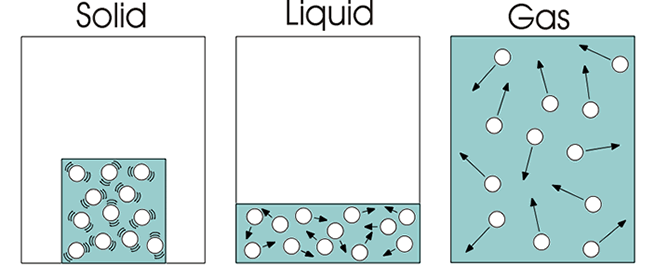
I'm using the same potential energy diagram to describe the energetics of gases. Because the gas has no lattice or anything which breaks rotational symmetry, you might think that on average, the volume that each particle occupies is a sphere surrounding that particle.  
In a gas, there is not enough attraction to stop the moving away from one another. They fall back towards one another and the process repeats. At this point, there will not be enough KE to keep moving away from one another. This will happen at a distance at the distance d. Well the amount of work to stop them will be equal to their KEs. The attractive force will apply itself across some distance. They are now moving away, each with some KE. Assuming the particles then “collide,” elastically of course, the particles reverse directions. In other words, all of the PE is converted to KE. The amount of work (or force * dist) to bring the particle to a certain velocity is its kinetic energy. The attractive force is applied across the distance between them, hence work. Since they attract each other, they fall towards one another. We can define the PE to be 0 at this distance, d. Assume the particles are at some finite distance from each other, each with no KE. For the following, assume two particles in one dimension. This also has to be why solids vibrate in place. This is my rationalization of the answer. Thought I would post this in case anyone else was confused. 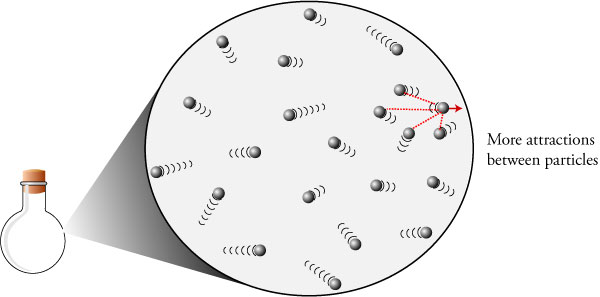
Two particles that are attracted to one another in close proximity would have a positive potential energy, right? How do we know that the kinetic energy term has to be greater than the potential energy term? On the other hand, the tendency for the particles to attract is represented by a potential energy term. The characteristic distance between the molecules. This motion is where the kinetic energy term comes from. particles, the impacts of individual particles on the walls merge into time-independent pressure. However, I don't quite understand the energetics of such a situation. The particles have are moving very quickly which trumps any attractive forces. Table 2 gives some typical values."In the gas phase, the molecules are freely moving particles traveling through space, where the kinetic energy associated with each particle is greater than the potential energy of intermolecular forces." 'In the gas phase, the molecules are freely moving particles traveling through space, where the kinetic energy associated with each particle is greater than the potential energy of intermolecular forces.' Qualitatively, this makes perfect sense. will take the cube root of the volume per molecule as an In a 1-M solution, there are 6×10^23 molecules/l, = 0.6 molecules/nm^3, or inverting, the volume per molecule is V=1.66 nm^3/molecule at 1 M. "It is frequently useful to know the average volume of solution occupiedīy each molecule, or more directly, the average distance Size and shape of protein molecules at the nanometer level determined by sedimentation, gel filtration, and electron microscopy. Distance between molecules as function of concentration RangeĮrickson HP. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
